Healthcare workers can be exposed to biological fluids that are capable of transmitting diseases. Those diseases, which are caused by a variety of microorganisms such as, Hepatitis B virus (HBV), Hepatitis C virus (HCV), Ebola Virus, and Human Immunodeficiency Virus (HIV) can pose significant risks to life and health. Healthcare workers wear protective clothing (e.g., surgical gowns, isolation gowns, and coveralls) to protect both patients and themselves from the transfer of microorganisms by blood and body fluids. A common misunderstanding among many end users is that they are protected from blood, body fluids, and other potentially infectious materials when they wear any type of fluid-resistant garment.
A common misunderstanding among many end users is that they are protected from blood, body fluids, and other potentially infectious materials when they wear any type of fluid-resistant garment. This document provides an overview of scientific evidence and information on national and international standards, test methods, and specifications for fluid-resistant and impermeable gowns and coveralls used in healthcare. This document focuses on selecting protective clothing primarily on the basis of their barrier properties.
The key to proper selection and use of gowns and coveralls is to understand the hazards and the risk of exposure.
The Centers for Disease Control and Prevention (CDC) has categorized three primary routes of transmission:
- Contact (direct and indirect).
- Respiratory droplets.
- Airborne droplet nuclei
By using appropriate protective clothing, it is possible to create a barrier to eliminate or reduce contact and droplet exposure, and therefore prevent the transfer of microorganisms between patients and healthcare workers.
Once the hazard and the risks of exposure are identified, gown and coverall selection can be guided by current scientific understanding of how protective clothing materials provide protection against microorganisms in blood and body fluids. A microorganism’s movement through protective clothing materials depends upon several factors, including the following:
Physical and chemical properties of the fabric: Includes factors such as thickness pore size, and repellency
Shape, size, and other characteristics of the microorganisms: Includes factors such as morphology, motility, and adaptation to environmental extremes
Characteristics of the carriers: Includes factors such as surface tension, volume, and viscosity
External factors: Includes factors such as physical, chemical, and thermal stresses
Several different microorganisms have been found in healthcare settings, including bacteria, viruses, and some fungi. The shape and size of microorganisms varies, and this will affect their ability to move through a fabric structure. In general, fungi are larger than bacteria, and bacteria are larger than viruses. For instance, HIV virus is spherical and 100–120 nanometers (nm) in diameter. The Ebola virus is a single-stranded RNA virus with a filamentous shape, a median particle length ranging from 974 nm to 1,086 nm, and average 80 nm in diameter.
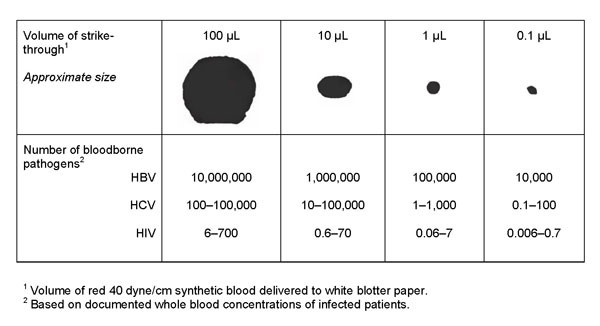
Microorganisms are transported by carriers such as body fluids, sloughed skin cells, lint, dust, and respiratory droplets. A significant number of microorganisms can be carried in a very minute volume of blood or body fluids, which may not be visible to the naked eye
American National Standard for Limited-Use and Disposable Coveralls—Size and Labeling Requirements,” includes a sizing chart and a set of exercises in which a user can validate that a garment is the proper size, thereby assisting facilities in selecting the appropriate size for each wearer.
Current Healthcare Protective Clothing Standards and Specifications
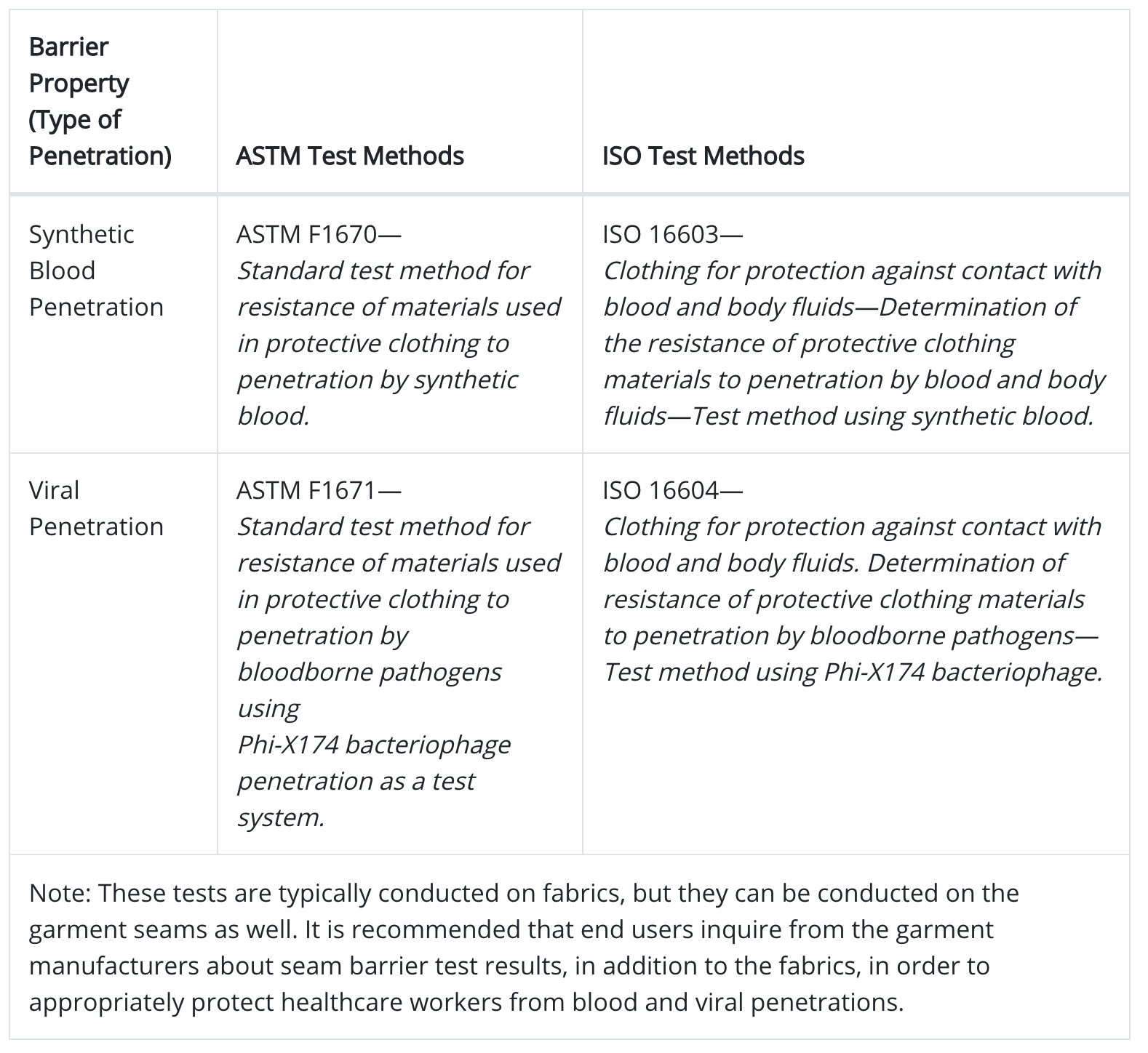
When the transmission route is defined as “direct contact transmission,” such as in the case of Covid 19 and HIV, employers should consider gowns and coveralls that demonstrate resistance to synthetic blood, as well as passage of virus. Standard test methods can be used to evaluate the resistance of fabrics or seams/closures to synthetic blood penetration and viral penetration, as described in Table 1.
The United States commonly uses American Society of Testing and Materials International (ASTM) methods, while Europe commonly uses International Organization for Standardization (ISO) methods
Table 1:
In the United States, ANSI/AAMI PB70 establishes a system of classification for protective apparel (including surgical gowns and isolation gowns) used in healthcare facilities, based on their liquid barrier performance. It also specifies labeling requirements and test methods for determining the compliance of protective apparel labeled with liquid barrier claims or liquid-borne microbial barrier claims. The ANSI/AAMI PB70 was accepted by the FDA in 2004.
The ANSI/AAMI PB70 standard includes four standard tests to evaluate the barrier effectiveness of surgical gowns, isolation gowns, and surgical drapes. Based on the results of these standardized tests, four levels of barrier performance are defined, with Level 1 being the lowest level of protection, and Level 4 being the highest level of protection. Table 3 summarizes the requirements of ANSI/AAMI PB70:2012 regarding the classification of barrier performance of surgical gowns, isolation gowns, and surgical drapes.
Table 2: ANSI/AAMI PB 70:12 classification of barrier performance of surgical gowns, other protective apparel, surgical drapes and drape accessories.

As indicated in Table 2, the requirements for levels 1, 2, and 3 have specific test requirements associated with them. Only Level 4 gowns are tested for viral penetration resistance, and therefore only Level 4 garments are considered impermeable to viral penetration using ASTM F1671. The gowns complying with the lower levels (Level 1, 2, and 3) cannot be considered impermeable. However, Level 1–3 gowns can provide increasing resistance to liquids. ANSI/AAMI PB70 Level 1, 2, and 3 surgical/isolation gown-testing requirements only use water as a challenge. Because the surface tension of water is much higher than that of blood, blood can penetrate through fabrics more readily than water. Therefore, no correlation can be made between AATCC water resistance tests (AATCC 42 and AATCC 127) and ASTM F1671 viral penetration tests, and consequently, protection provided by Levels 1, 2, 3 gowns and that of Level 4 gowns. A common misunderstanding among many endusers is that they are protected from blood, body fluids, and other potentially infectious materials (OPIM) when they wear any type of fluid-resistant garment or surgical or isolation gown.
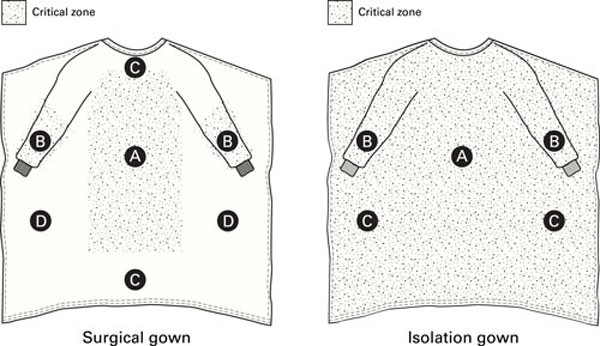
The requirements for the design and construction of surgical and isolation gowns are based on the anticipated location and degree of liquid contact, given the expected conditions of use. ANSI/AAMI PB70:2012 identifies certain areas of surgical and isolation gowns as critical zones (see Figure 3). [ANSI 2012] The critical zones include those areas where direct contact with blood, body fluids, and/or OPIM is most likely to occur—even though the areas outside of critical zones can inadvertently be splashed or sprayed as well. According to the standard, for isolation gowns the whole garment is considered a critical zone due to the unpredictable types of potential contact with blood, body fluids, and OPIM. The entire isolation gown, including the seams, but excluding the cuffs, hems, and bindings, must achieve claimed barrier performance. Open-backed isolation gowns do not meet the critical area parameters, and therefore they cannot be rated. For surgical gowns, the critical zone comprises at least the front panel (area A) and lower sleeves (area B) (Figure 3). The classification of the surgical gown is based on the lower performing component of the two. According to the standard, the back of the gown (area D) can be non-protective. However, the entire front of the gown (areas A, B, and C) is required to have a barrier performance of at least Level 1. Therefore, the areas outside of critical areas in the front of gown (i.e., area C) can be Level 1 even though the whole gown is classified as Level 4 surgical gown. Therefore, using a surgical gown in isolation settings may not provide appropriate protection.
Figure 3: Critical zones defined for surgical gowns and isolation gowns in ANSI/AAMI PB70. Adapted with permission from ANSI/AAMI PB70:2012, “Liquid barrier performance and classification of protective apparel and drapes intended for use in health care facilities.”
In addition to ANSI/AAMI PB70 barrier resistance testing requirements, this standard specification also establishes physical property and comfort requirements including tensile strength, seam strength, tear resistance, lint generation, evaporative resistance, and moisture vapor transmission rate, mainly for reporting purposes only [ASTM 2407 2006].
EN 13795—“Surgical drapes, gowns and clean air suits, used as medical devices for patients, clinical staff and equipment. General requirements for manufacturers, processors and products, test methods, performance requirements and performance levels.”
EN 13795 is a European recognized standard of quality and conformance to manufacturing, testing and performance specifications for single-use and multiple-use surgical gowns, drapes, and clean-air suits. The standard was developed to assist healthcare professionals in making informed decisions when selecting the appropriate product for the anticipated tasks by specifying a consistent basis for testing and providing a common understanding of barrier properties. EN 13795 categorizes products by performance type: high performance versus standard performance gown classes. It then further subdivides them into critical and less critical product areas as in ANSI/AAMI PB70 (critical zones in surgical gowns).
EN 13795 also describes the standardized and harmonized barrier test methodologies that single-use and multiple-use surgical gowns must undergo. Table shows EN 13795 specific performance requirements for surgical gowns. In addition to these tests, EN 13795 requires other characteristics be tested, including microbial and particulate matter cleanliness, linting, bursting strength (dry and wet), and tensile strength (dry and wet). However, performance requirements for these characteristics are the same for both standard-performance and high-performance surgical gowns, and critical and less-critical areas.
Note that EN 13795 is aimed to protect the patient; therefore, within the context of the European standard, the products conforming to this standard are considered medical devices. If a product is designed to be PPE rather than a medical device, the product has to comply with the corresponding directive 89/686/EEC (protective clothing) and standard “EN 14126 Performance requirements and tests methods for protective clothing against infective agents.” EN 14126 has various classes for most performance requirements; therefore, if EN 14126 is specified, then the classes for the performance requirements should also be specified.
Table 3. Overview of some of the EN 13795 performance requirements
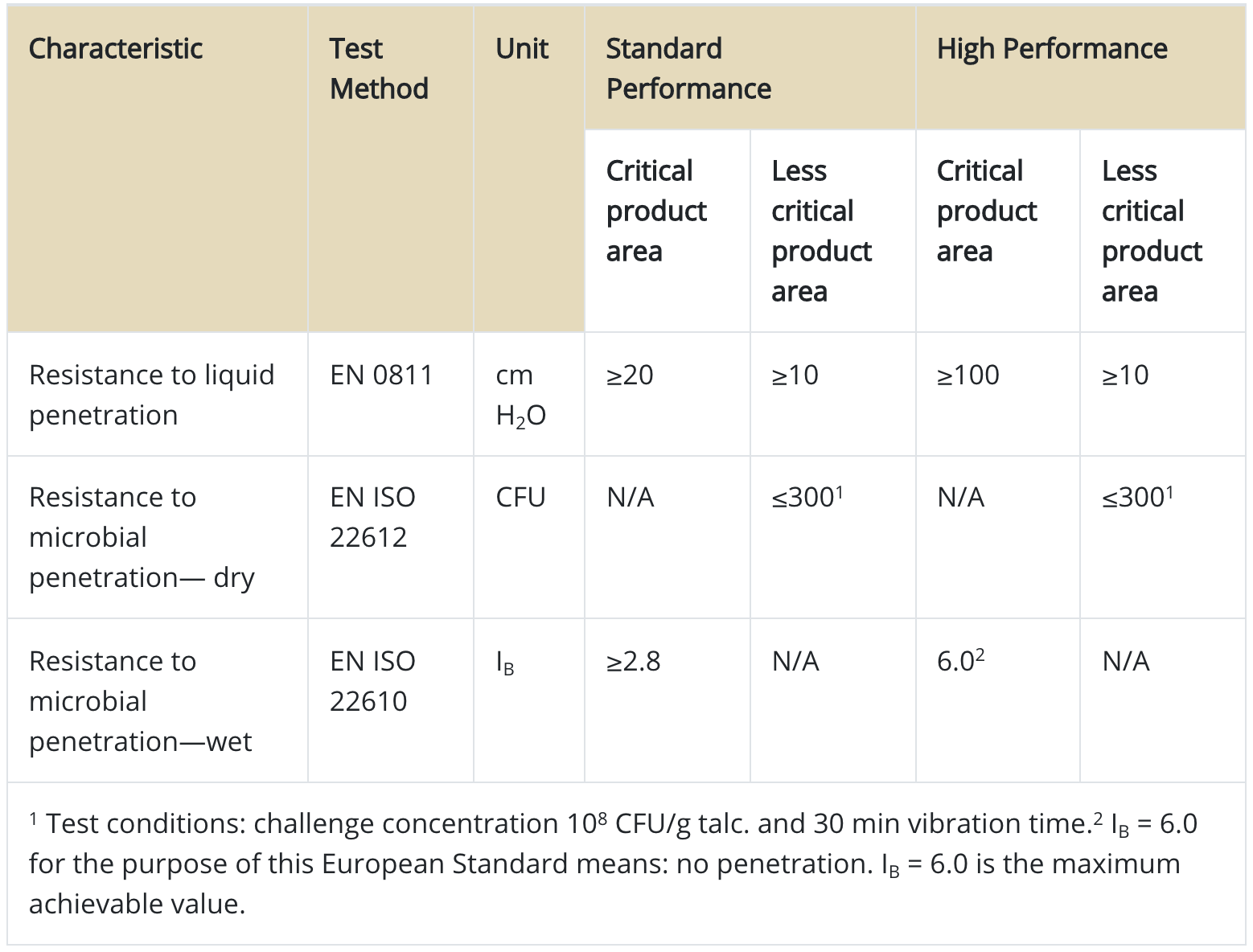
Similar to “AATCC 127, Water resistance: hydrostatic pressure test,” “EN 20811 Textiles. Determination of resistance to water penetration. Hydrostatic pressure test” evaluates the behavior of the fabric structure under constantly increasing hydrostatic pressure. The results are expressed in water column (cm), and higher values indicate a better resistance. Note that ANSI/AAMI PB70 also cites this method (AATCC 127) for one of the classification requirements.
The EN ISO 22612 test method for resistance to dry microbial penetration is designed to determine the ability of dry fabric to resist penetration of particles carrying microorganisms. The method establishes the quantity of microorganisms that can penetrate through the test material while being carried on talcum powder. Test results are expressed in colony forming units (CFU) observed on the agar plate.
The EN ISO 22610 test method, used to determine the resistance to wet bacterial penetration, evaluates fabric’s resistance to microbial penetration under conditions of liquid pooling on the fabric and mechanical rubbing. Test results are expressed in IB “Barrier Index.” IB=6.0 is the maximum achievable value, and it means no penetration for the purpose of the test.
EN 14126—“Performance requirements and tests methods for protective clothing against infective agents.”
The European standard EN 14126 defines performance requirements for materials in protective clothing used to protect from infectious agents. Due to the heterogeneity of microorganisms (in terms of size, shape, infectious dose, survival abilities, etc.), the EN 14126 standard does not define performance criteria for specific types of microorganisms. The test methods specified in this standard focus on the medium containing the microorganism, such as liquid, aerosol, or solid dust particle.
The EN 14126 standard is typically used for coveralls and it specifies ISO 16603 and ISO 16604 as test methods used to evaluate the penetration resistance performance of clothing materials to contaminated liquids under hydrostatic pressure. Clothing materials are classified based on the performance levels achieved by these test methods (see Table 5). Note that the pressure level in Class 5 is closest to the pressure levels specified in ASTM F1670 and ASTM F1671 (13.8 kPa).
Note that EN 14126 specifications for ISO 16603 and ISO 16604 apply only to fabrics used in the garments. Both tests could be conducted on the seams as well. In order to be appropriately protected from blood and viral penetrations, end users should consult garment manufacturers about the seam barrier test results, in addition to the fabric test results.
Table 4: Classification according to EN 14126 of resistance to penetration by blood and body fluids using ISO 16603 and ISO 16604 test methods

Several test methods and performance requirements for barrier resistant gowns and coveralls were discussed in this technical document. Test methods for determination of the barrier resistance of fabrics such as AATCC 42, AATCC 127, ASTM F1670, ASTM F1671, ISO 16603, ISO 16604, are used for both coveralls and gowns. In general, for the classification of the protective clothing, ANSI/AAMI PB70 and EN 13795 are used for gowns and EN 14126 and NFPA 1999 are used for coveralls.
There are several differences between ANSI/AAMI PB70 and EN 13795 surgical gown classifications. Because the test methods and performance requirements cannot be compared directly, it is difficult to assign equivalency between surgical gowns classified according to ANSI/AAMI PB70 (see Table 3) and EN 13795 (see Table 4). Recent PPE specifications from the World Health Organizationpdf iconexternal icon for Filovirus disease outbreak indicate that EN 13795 high performance level gown is most similar to ANSI/AAMI PB70 level 3 gown.
Similarly, for coveralls it is difficult to compare test methods and performance specifications directly. In Europe, the EN 14126 standard typically is used to evaluate and classify coveralls used to protect from infectious agents and EN 13795 is used to evaluate and classify surgical gowns. Unlike surgical or isolation gowns (ANSI/AAMI PB70), there is no widely used classification standard in the United States. Coveralls with materials and seams tested against ASTM 1671 are specified in NFPA 1999–2013, Standard on Protective Clothing for Emergency Medical Operations. This standard establishes minimum performance requirements for single-use emergency medical garments, multiple-use emergency medical garments, and other PPE for protection from contact with blood and body-fluid-borne pathogens for personnel performing patient care during emergency medical operations [NFPA 2013]. While originally designed for pre-hospital healthcare workers, it could be used for hospital-based healthcare workers as well.
Thus, the current best approach to comparing coveralls is to use manufacturer supplied test data. Many of the same test methods used to estimate barrier protection of gowns (Tables 3 and 4) can be used for coveralls as well. Table 6 lists some of the commonly used test methods for determining the barrier effectiveness of coveralls and describes how the results should be interpreted.

